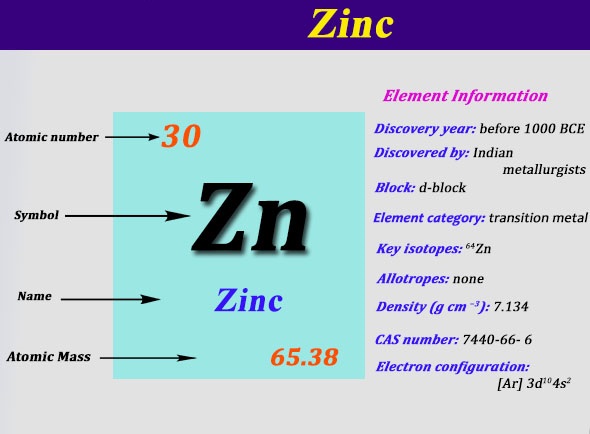 
When people absorb too little zinc they can experience a loss of appetite, decreased sense of taste and smell, slow wound healing and skin sores. Zinc is a trace element that is essential for human health. More than 30% of the world's need for zinc is met by recycling. World production exceeds 7 million tonnes a year and commercially exploitable reserves exceed 100 million tonnes. The main zinc mining areas are Canada, Russia, Australia, USA and Peru'. Other important zinc ores are wurzite, smithsonite and hemimorphite. The dominant ore is zinc blende, also known as sphalerite. Zinc is the 23rd most abundant element in the Earth's crust. Some soils are heavily contaminated with zinc, and these are to be found in areas where zinc has to be mined or refined, or were sewage sludge from industrial areas has been used as fertilizer. Most zinc is added during industrial activities, such as mining, coal and waste combustion and steel processing. Zinc occurs naturally in air, water and soil, but zinc concentrations are rising unnaturally, due to addition of zinc through human activities. Industrial sources or toxic waste sites may cause the zinc amounts in drinking water to reach levels that can cause health problems. Drinking water also contains certain amounts of zinc, which may be higher when it is stored in metal tanks. Many foodstuffs contain certain concentrations of zinc. Zinc is a very common substance that occurs naturally. Zinc metal is included in most single tablet, it is believed to possess anti-oxidant properties, which protect against premature aging of the skin and muscles of the body. As a pigment zinc is used in plastics, cosmetics, photocopier paper, wallpaper, printing inks etc, while in rubber production its role is to act as a catalyst during manufacture and as a heat disperser in the final product. Zinc oxide is used as a white pigment in watercolours or paints, and as an activator in the rubber industry. Zinc is the primary metal used in making American pennies, is used in die casting in the automobile industry. It is used for the negative plates in some electric batteries and for roofing and gutters in building construction. It is used principally for galvanizing iron, more than 50% of metallic zinc goes into galvanizing steel, but is also important in the preparation of certain alloys. It is a fairly reactive metal that will combine with oxygen and other non-metals, and will react with dilute acids to release hydrogen. It is brittle and crystalline at ordinary temperatures, but it becomes ductile and malleable when heated between 110☌ and 150☌. It is found in group IIb of the periodic table. Zinc - Zn Chemical properties of zinc - Health effects of zinc - Environmental effects of zinc Separation and Concentration Purification Request.Plant Inspection & Process Optimalisation.Zinc carbonate and zinc gluconate are used as dietary supplements, zinc chloride in deodorants), zinc pyrithione (anti-dandruff shampoos) and dimethylzinc in the organic laboratory.Other applications of zinc include electrical batteries and small non-structural castings.Brass, a zinc alloy is useful in making communication equipment, hardware, musical instruments, and water valves.Galvanized steel is used in manufacturing vehicles. Zinc is also used in galvanizing iron and steel as it prevents iron from rusting.Zinc sulphide (ZnS) is used in luminescent pigments such as on the hands of clocks, X-rays and television screens.Other compounds of zinc are used in model rockets, salting material for nuclear weapons, and fire retardants.It protects rubber polymers and plastics from ultraviolet radiation. It is used in paints and as a catalyst in the manufacture of rubber to disperse heat. Zinc is a widely used metal across various sectors.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
